 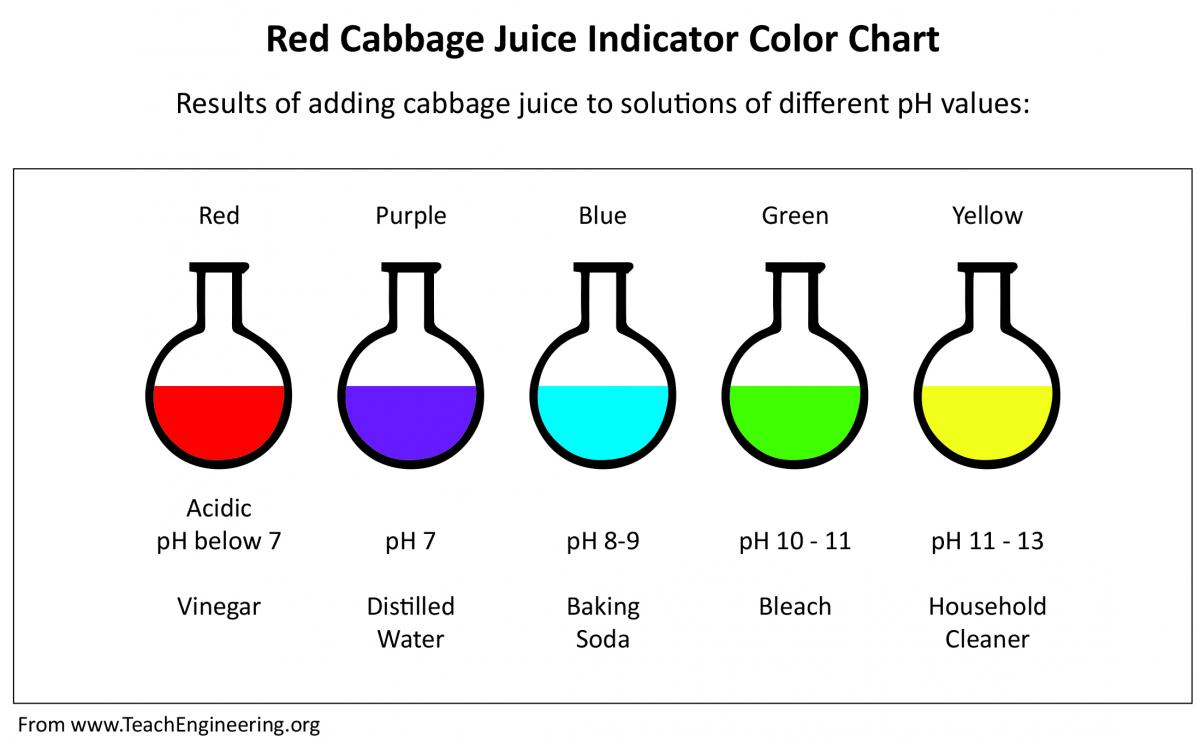 
An indicator is a substance that changes color when you add an acid or base, and can determine the acidity or basicity of a substance visually based on the color it changes to. One method of figuring out if a substance is an acid or a base is by using a pH indicator. A neutral solution on the scale has a pH of 7, any solution lower than 7 is considered an acid, and any solution higher than 7 is considered a base. The pH of a substance expresses the acidity or basicity of a solution by using a scale that measures from 0 to 14. Both acids and bases can also be classified by identifying the pH of the solution. The chemical properties of bases are: that aqueous solutions of bases taste bitter, bases change the color of acid-base indicators, bases feel slippery, bases react with acids to produce salts and waters, and bases conduct an electric current. The chemical properties of acids are: that aqueous solutions of acids taste sour, acids change the color of acid-base indicators, some acids react with metals to form hydrogen gas (H 2 ), acids react with bases to produce salts and water, and acids conduct an electric current. Acids and bases have different properties that can help in classifying what type a solution is. The substances in this experiment can be categorized into two sections which are acids and bases. The tested chemicals used in this experiment may be safely washed down the drain with water when performing the last step in this experiment. If any acid or base does come to contact with skin make sure to wash the affected area thoroughly with water to dilute the substance. The use of gloves serve as a precaution when handling acids or bases to prevent any substance from coming to contact with the skin. Throughout this experiment there will be the use of weak acids and bases.Weak acids and bases are not as corrosive as strong acids or bases, but are capable of causing damage to the skin if left in area for a long period. When cleaning the blender be aware of the blades and clean it mindfully to avoid any cuts. If there is any smell of burning coming from the blender immediately unplug the blender and remove the food. Make sure not to run the blender for a long time because this can cause a short circuit. To prevent any spilling or accident make sure to always keep the lid on when using the blender. When using the blender in this experiment make sure to fill it with the ingredients first before you turn it on. Use a pH meter to record the actual pH of each substance.Record predicted pH from the cabbage juice color scale of each substance tested.Add test chemicals to the cabbage juice and stir until there’s a color change.Fill each cup with 140 mL of cabbage juice.Peel off 3-4 big cabbage leaves and put them in the blender.Test Chemicals: Apple cider vinegar (CH 3 COOH), baking soda (NaHCO 3 ), lemon juice (C 6 H 8 O 7 ), laundry detergent (KOH),ammonia (NH 3 ), and soda (H 2 CO 3 ).PH scale for Red Cabbage juice, stirring, same amount of red cabbage juice, clear drinking glasses, pH meter. The color when an acid or base is added to the red cabbage indicator. The different substances tested: ammonia (NH 3 ), baking soda (NaHCO 3 ), apple cider vinegar (CH 3 COOH), lemon juice (C 6 H 8 O 7 ), soda pop (H 2 CO 3 ), and laundry detergent (KOH). Using a pH color scale for a red cabbage juice indicator I will predict the the approximate pH of common household liquids and items such as: ammonia, baking soda, apple cider vinegar, lemon juice, soda pop, and laundry detergent by observing the change in color, and comparing it to the actual pH. Is Red Cabbage juice an accurate pH indicator?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
